Fabrication and Characterization Fe3O4/Humic Acid for the Efficient Removal of Malachite Green
Abstract
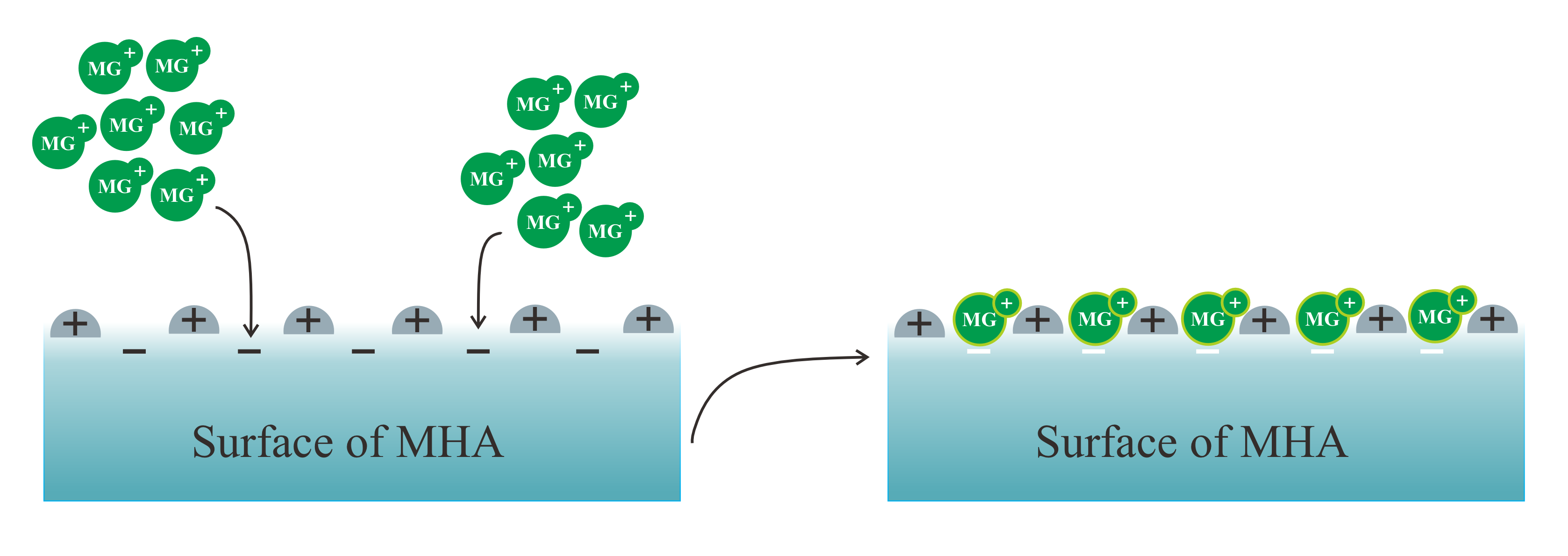
The dye pollutants that contaminate water and food resulting from commercial manufacture and illicit addiction are a worldwide threat that harms the ecosystem, the food supply, and the health of humans. Magnetite/Fe3O4 humic acid (MHA) with various ratios was synthesized using a two-step process involving coprecipitation and hydrothermal treatment in order to effectively overcome these obstacles. Analyses of SEM, XRD, FTIR, and VSM were used to describe the morphology and physicochemical aspects of MHA. The adsorption kinetics studies indicated that the adsorption mechanism of malachite green adhered to the pseudo-second-model and that the adsorption was adequately described by the Langmuir isotherm. The thermodynamic studies demonstrated spontaneous, endothermic, disorderly adsorption. MHA2 had maximal malachite green adsorption capacities of 83.333 mg/g. Malachite green and MHA may interact via interaction, electrostatic attraction, van der Waals forces, H-bonding, pore filling, pore locking, and/or mechanical adhesion; however, physisorption dominates the adsorption process. Malachite green's adsorption characteristics change significantly after up to four cycles. It has been demonstrated that MHA has a high capacity for dye adsorption and a broad range of potential applications.
References
Ahmad, N., Suryani Arsyad, F., Royani, I., Mega Syah Bahar Nur Siregar, P., Taher, T., & Lesbani, A. (2023). High regeneration of ZnAl/NiAl-Magnetite humic acid for adsorption of Congo red from aqueous solution. Inorganic Chemistry Communications, 150.
Ahmed, S., Rehman, H. U., Ali, Z., Qadeer, A., Haseeb, A., & Ajmal, Z. (2021). Solvent assisted synthesis of hierarchical magnesium oxide flowers for adsorption of phosphate and methyl orange: Kinetic, isotherm, thermodynamic and removal mechanism. Surfaces and Interfaces, 23(May 2020), 100953.
Altun, T., & Ecevit, H. (2022). Adsorption of malachite green and methyl violet 2B by halloysite nanotube: Batch adsorption experiments and Box-Behnken experimental design. Materials Chemistry and Physics, 291.
Amiri, M., Salavati-Niasari, M., Akbari, A., & Gholami, T. (2017). Removal of malachite green (a toxic dye) from water by cobalt ferrite silica magnetic nanocomposite: Herbal and green sol-gel autocombustion synthesis. International Journal of Hydrogen Energy, 42(39), 24846–24860.
Bakhshi Nejad, S., & Mohammadi, A. (2020). Epoxy-Triazinetrione-Functionalized Magnetic Nanoparticles as an Efficient Magnetic Nanoadsorbent for the Removal of Malachite Green and Pb(II) from Aqueous Solutions. Journal of Chemical and Engineering Data, 65(5), 2731–2742.
Ben, S. K., Gupta, S., Raj, K. K., & Chandra, V. (2023). Adsorption of malachite green from polyaniline facilitated cobalt phosphate nanocomposite from aqueous solution. Chemical Physics Letters, 820.
Brahma, D., Nath, H., Borah, D., Debnath, M., & Saikia, H. (2022). Coconut Husk Ash Fabricated CoAl-Layered Double Hydroxide Composite for the Enhanced Sorption of Malachite Green Dye: Isotherm, kinetics and thermodynamic studies. Inorganic Chemistry Communications, 144.
Cardinale, A. M., Carbone, C., Fortunato, M., Fabiano, B., & Reverberi, A. Pietro. (2022). ZnAl-SO4 Layered Double Hydroxide and Allophane for Cr(VI), Cu(II) and Fe(III) Adsorption in Wastewater: Structure Comparison and Synergistic Effects. Materials, 15(19).
Cheng, C. M., Patel, A. K., Singhania, R. R., Tsai, C. H., Chen, S. Y., Chen, C. W., & Dong, C. Di. (2021). Heterologous expression of bacterial CotA-laccase, characterization and its application for biodegradation of malachite green. Bioresource Technology, 340.
Dahlan, I., Keat, O. H., Aziz, H. A., & Hung, Y. T. (2023). Synthesis and characterization of MOF-5 incorporated waste-derived siliceous materials for the removal of malachite green dye from aqueous solution. Sustainable Chemistry and Pharmacy, 31.
Das, K. C., & Dhar, S. S. (2020). Rapid catalytic degradation of malachite green by MgFe2O4 nanoparticles in presence of H2O2. Journal of Alloys and Compounds, 828, 154462.
Fagbohun, E. O., Wang, Q., Spessato, L., Zheng, Y., Li, W., Ganiyat, O. A., & Cui, Y. (2021). Physicochemical regeneration of industrial spent activated carbons using a green activating agent and their adsorption for methyl orange. Surfaces and Interfaces, 29(December 2021), 101696.
Faiza Amin, K., Gulshan, F., Asrafuzzaman, Das, H., Rashid, R., & Manjura Hoque, S. (2023). Synthesis of mesoporous silica and chitosan-coated magnetite nanoparticles for heavy metal adsorption from wastewater. Environmental Nanotechnology, Monitoring & Management, 100801.
Fan, S., Fan, X., Wang, S., Li, B., Zhou, N., & Xu, H. (2023). Effect of chitosan modification on the properties of magnetic porous biochar and its adsorption performance towards tetracycline and Cu2+. Sustainable Chemistry and Pharmacy, 33, 101057.
Gao, L., Gao, T., Zhang, Y., & Hu, T. (2022). A bifunctional 3D porous Zn-MOF: Fluorescence recognition of Fe3+ and adsorption of congo red/methyl orange dyes in aqueous medium. Dyes and Pigments, 197(November 2021), 109945.
Gautam, R. K., Rawat, V., Banerjee, S., Sanroman, M. A., Soni, S., Singh, S. K., & Chattopadhyaya, M. C. (2015). Synthesis of bimetallic Fe-Zn nanoparticles and its application towards adsorptive removal of carcinogenic dye malachite green and Congo red in water. Journal of Molecular Liquids, 212, 227–236.
Gong, L., Wu, H., Shan, X., & Li, Z. (2021). Facile fabrication of phosphorylated alkali lignin microparticles for efficient adsorption of antibiotics and heavy metal ions in water. Journal of Environmental Chemical Engineering, 9(6).
Grover, A., Mohiuddin, I., Lee, J., Brown, R. J. C., Malik, A. K., Aulakh, J. S., & Kim, K. H. (2022). Progress in pre-treatment and extraction of organic and inorganic pollutants by layered double hydroxide for trace-level analysis. In Environmental Research (Vol. 214). Academic Press Inc.
Hoang, L. P., Van, H. T., Hang Nguyen, T. T., Nguyen, V. Q., & Thang, P. Q. (2020). Coconut shell activated carbon/CoFe2O4 composite for the removal of rhodamine B from aqueous solution. Journal of Chemistry, 2020.
Jabeen, S., Ganie, A. S., Ahmad, N., Hijazi, S., Bala, S., Bano, D., & Khan, T. (2023). Fabrication and studies of LaFe2O3/Sb2O3 heterojunction for enhanced degradation of Malachite green dye under visible light irradiation. Inorganic Chemistry Communications, 152.
Jin, C., Liu, Y., Fan, J., Liu, T., Liu, G., Chu, F., & Kong, Z. (2022). Lignin-inspired porous polymer networks as high-performance adsorbents for the efficient removal of malachite green dye. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 643.
Kavci, E. (2021). Malachite green adsorption onto modified pine cone: Isotherms, kinetics and thermodynamics mechanism. Chemical Engineering Communications, 208(3), 318–327.
Khan, A. N., & Bagla, H. K. (2022). Batch adsorption and desorption investigations of Cs(I) and Sr(II) from simulated reactor waste by humic acid. Journal of Trace Elements and Minerals, 1, 100005.
Kotnala, S., Bhushan, B., & Nayak, A. (2023). Fabrication of a magnetite hydroxyapatite nanocomposite for the removal of Paraquat dichloride: Adsorption studies. Materials Today: Proceedings, 73, 122–127.
Lee, W.-H., & Kim, J.-O. (2022). Phosphate recovery from anaerobic digestion effluent using synthetic magnetite particles. Journal of Environmental Chemical Engineering, 10(1), 107103.
Leng, L., Yuan, X., Zeng, G., Shao, J., Chen, X., Wu, Z., Wang, H., & Peng, X. (2015). Surface characterization of rice husk bio-char produced by liquefaction and application for cationic dye (Malachite green) adsorption. Fuel, 155, 77–85.
Li, D., Hua, T., Yuan, J., & Xu, F. (2021). Methylene blue adsorption from an aqueous solution by a magnetic graphene oxide/humic acid composite. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 627.
Li, H., Yuan, Z., Ding, S., & Yuan, J. (2023). Adsorption of lead ions by magnetic carbon: Comparison of magnetic carbon properties and modification methods. Journal of Environmental Chemical Engineering, 11(3).
Mohammed Al-Balawi, A., Zaheer, Z., & Kosa, S. A. (2023). Silver-platinum bimetallic nanoparticles as heterogeneous persulfate activator for the oxidation of malachite green. Arabian Journal of Chemistry, 16(8).
Muinde, V. M., Onyari, J. M., Wamalwa, B., & Wabomba, J. N. (2020a). Adsorption of malachite green dye from aqueous solutions using mesoporous chitosan–zinc oxide composite material. Environmental Chemistry and Ecotoxicology, 2, 115–125.
Muinde, V. M., Onyari, J. M., Wamalwa, B., & Wabomba, J. N. (2020b). Adsorption of malachite green dye from aqueous solutions using mesoporous chitosan–zinc oxide composite material. Environmental Chemistry and Ecotoxicology, 2, 115–125.
Palapa, N. R., Ahmad, N., Wijaya, A., & Zahara, Z. A. (2023). Facile Fabrication of Layered Double Hydroxide-Lignin for Efficient Adsorption of Malachite Green. Science and Technology Indonesia, 8(2), 305–311.
Paz, M. J., Vieira, T., Enzweiler, H., & Paulino, A. T. (2022). Chitosan/wood sawdust/magnetite composite membranes for the photodegradation of agrochemicals in water. Journal of Environmental Chemical Engineering, 10(1), 106967.
Prilepskii, A. Y., Fakhardo, A. F., Drozdov, A. S., Vinogradov, V. V., Dudanov, I. P., Shtil, A. A., Bel’Tyukov, P. P., Shibeko, A. M., Koltsova, E. M., Nechipurenko, D. Y., & Vinogradov, V. V. (2018). Urokinase-Conjugated Magnetite Nanoparticles as a Promising Drug Delivery System for Targeted Thrombolysis: Synthesis and Preclinical Evaluation. ACS Applied Materials and Interfaces, 10(43), 36764–36775.
Qu, W., Yuan, T., Yin, G., Xu, S., Zhang, Q., & Su, H. (2019). Effect of properties of activated carbon on malachite green adsorption. Fuel, 249, 45–53.
Rahman, M. T., Kameda, T., Miura, T., Kumagai, S., & Yoshioka, T. (2021). Removal of sulfate from wastewater via synthetic Mg–Al layered double hydroxide: An adsorption, kinetics, and thermodynamic study. Journal of the Indian Chemical Society, 98(11).
Rajabi, M., Mirza, B., Mahanpoor, K., Mirjalili, M., Najafi, F., Moradi, O., Sadegh, H., Shahryari-ghoshekandi, R., Asif, M., Tyagi, I., Agarwal, S., & Gupta, V. K. (2016). Adsorption of malachite green from aqueous solution by carboxylate group functionalized multi-walled carbon nanotubes: Determination of equilibrium and kinetics parameters. Journal of Industrial and Engineering Chemistry, 34, 130–138.
Raval, A. R., Kohli, H. P., & Mahadwad, O. K. (2022). Application of emulsion liquid membrane for removal of malachite green dye from aqueous solution: Extraction and stability studies. Chemical Engineering Journal Advances, 12.
Santosa, S. J., Krisbiantoro, P. A., Minh Ha, T. T., Thanh Phuong, N. T., & Gusrizal, G. (2021). Composite of magnetite and Zn/Al layered double hydroxide as a magnetically separable adsorbent for effective removal of humic acid. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 614(October 2020), 126159.
Sarkar, S., Tiwari, N., Basu, A., Behera, M., Das, B., Chakrabortty, S., Sanjay, K., Suar, M., Adhya, T. K., Banerjee, S., & Tripathy, S. K. (2021). Sorptive removal of malachite green from aqueous solution by magnetite/coir pith supported sodium alginate beads: Kinetics, isotherms, thermodynamics and parametric optimization. Environmental Technology and Innovation, 24.
Siraorarnroj, S., Kaewtrakulchai, N., Fuji, M., & Eiad-ua, A. (2022). High performance nanoporous carbon from mulberry leaves (Morus alba L.) residues via microwave treatment assisted hydrothermal-carbonization for methyl orange adsorption: Kinetic, equilibrium and thermodynamic studies. Materialia, 21(November 2021), 101288.
Sun, N., Liu, J., Qi, B. W., Lu, L. L., Du, H. L., Li, S., Li, C. Q., Jiang, S. W., Wang, Z. J., Yang, A. P., Zhu, G. L., Wang, T. Y., Wang, S. M., & Fu, Q. (2023). Effect of humic acid-modified attapulgite on polycyclic aromatic hydrocarbon adsorption and release from paddy soil into the overlying water in a rice-crab coculture paddy ecosystem and the underlying process. Chemosphere, 329.
Sun, Q., Zhou, H., Xu, C., Ba, Y., Geng, Z., & She, D. (2023). Effective adsorption of ammonium nitrogen by sulfonic-humic acid char and assessment of its recovery for application as nitrogen fertilizer. Science of the Total Environment, 867.
Tang, Y., Zhang, X., Li, X., Bai, J., Yang, C., Zhang, Y., Xu, Z., Jin, X., & Jiang, Y. (2023). Facile synthesis of magnetic ZnAl layered double hydroxides and efficient adsorption of malachite green and Congo red. Separation and Purification Technology, 322, 124305.
Tatarchuk, T., Soltys, L., & Macyk, W. (2023). Magnetic adsorbents for removal of pharmaceuticals: A review of adsorption properties. Journal of Molecular Liquids, 384, 122174.
Thotagamuge, R., Kooh, M. R. R., Mahadi, A. H., Lim, C. M., Abu, M., Jan, A., Hanipah, A. H. A., Khiong, Y. Y., & Shofry, A. (2021). Copper modified activated bamboo charcoal to enhance adsorption of heavy metals from industrial wastewater. Environmental Nanotechnology, Monitoring and Management, 16.
Tran, T. Van, Nguyen, D. T. C., Kumar, P. S., Din, A. T. M., Qazaq, A. S., & Vo, D. V. N. (2022). Green synthesis of Mn3O4 nanoparticles using Costus woodsonii flowers extract for effective removal of malachite green dye. Environmental Research, 214.
Wang, Q., Zhang, Y., Chen, H., Chen, S., & Wang, Y. (2023). Effects of humic acids on the adsorption of Pb(II) ions onto biofilm-developed microplastics in aqueous ecosystems. Science of the Total Environment, 882.
Wang, Y., Huang, Z., Sheng, L., & Ma, Y. (2023). Effect of modified humic acid residue on the adsorption and passivation of Hg2+/Pb2+ in solution and soil. Journal of Molecular Liquids, 377.
Wijaya, A., & Yuliasari, N. (2023). Biochar Derived from Rice Husk as Effective Adsorbent for the Removal Congo Red and Procion Red MX-5B Dyes. Indonesian Journal of Material Research, 1(1), 1–7.
Wu, F., Chen, L., Hu, P., Zhou, X., Zhou, H., Wang, D., Lu, X., & Mi, B. (2022). Comparison of properties, adsorption performance and mechanisms to Cd(II) on lignin-derived biochars under different pyrolysis temperatures by microwave heating. Environmental Technology and Innovation, 25.
Yan, C., Fan, L., Chen, Y., & Xiong, Y. (2020). Effective adsorption of oxytetracycline from aqueous solution by lanthanum modified magnetic humic acid. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 602.
Yönten, V., Sanyürek, N. K., & Kivanç, M. R. (2020). A thermodynamic and kinetic approach to adsorption of methyl orange from aqueous solution using a low cost activated carbon prepared from Vitis vinifera L. Surfaces and Interfaces, 20(February), 1–8.
Zeng, Y., Zhao, L., Wu, W., Lu, G., Xu, F., Tong, Y., Liu, W., & Du, J. (2013). Enhanced adsorption of malachite green onto carbon nanotube/polyaniline composites. Journal of Applied Polymer Science, 127(4), 2475–2482.
Zhang, J., Li, X., Xu, H., Zhang, W., Feng, X., Yao, Y., Ma, Y., Su, L., Ren, S., & Li, S. (2023). Removal of Cd2+, Pb2+ and Ni2+ from water by adsorption onto magnetic composites prepared using humic acid from waste biomass. Journal of Cleaner Production, 411.
Zhao, P., Wang, A., Wang, P., Huang, Z., Fu, Z., & Huang, Z. (2023). Two recyclable and complementary adsorbents of coal-based and bio-based humic acids: High efficient adsorption and immobilization remediation for Pb(II) contaminated water and soil. Chemosphere, 318.
Zhou, Y., Zhang, Y., Li, P., Li, G., & Jiang, T. (2014). Comparative study on the adsorption interactions of humic acid onto natural magnetite, hematite and quartz: Effect of initial HA concentration. Powder Technology, 251, 1–8.
Zong, E., Fan, R., Hua, H., Yang, J., Jiang, S., Dai, J., Liu, X., & Song, P. (2023). A magnetically recyclable lignin-based bio-adsorbent for efficient removal of Congo red from aqueous solution. International Journal of Biological Macromolecules, 226, 443–453.
Authors

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.