Enhanced Methods for Synthesizing Hydroxyxanthone Using Eaton and ZnCl2 Catalyst with Microwave Irradiation
Abstract
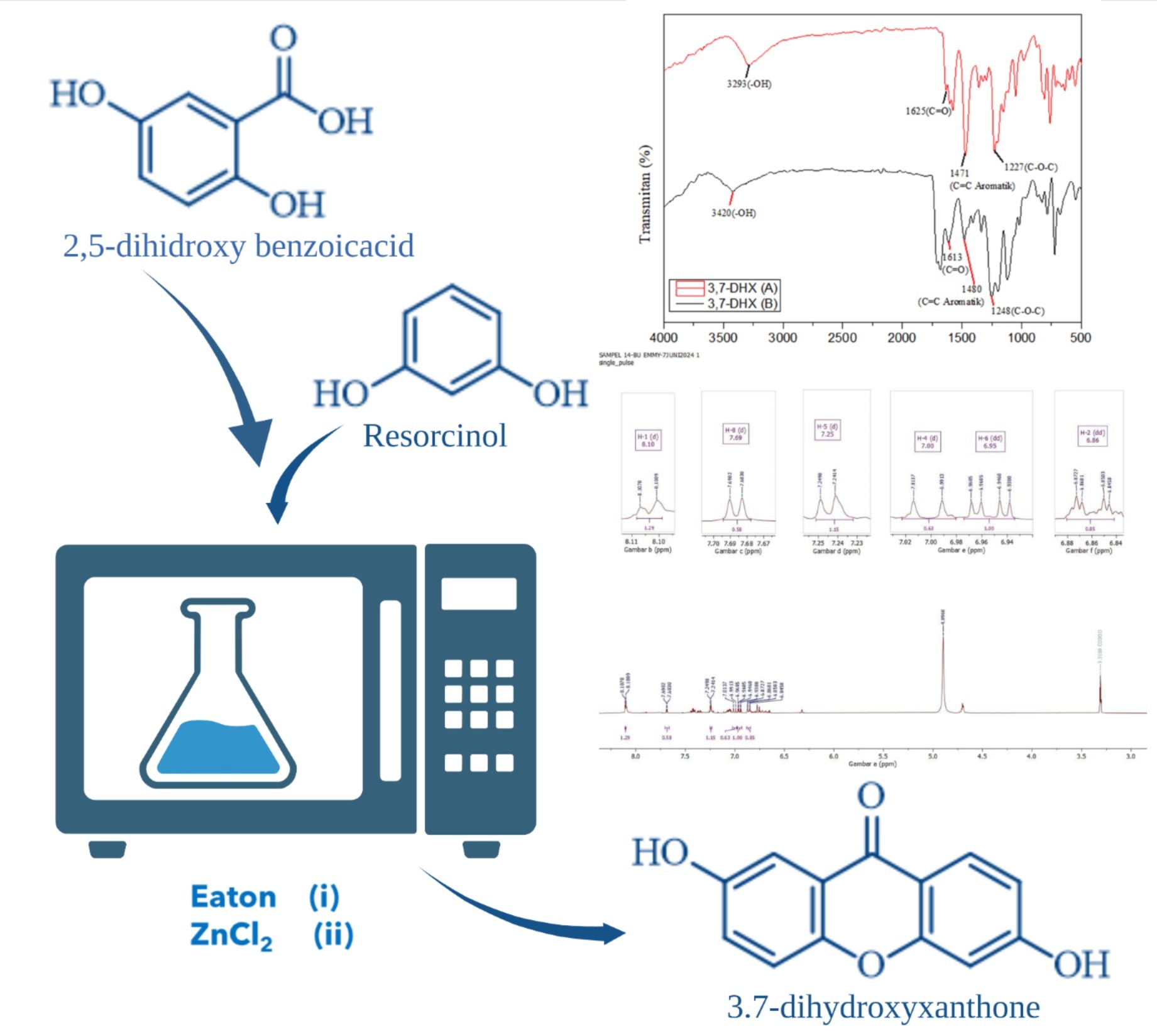
The methods for synthesizing hydroxyxanthone compounds are continually advancing. Among these, microwave-assisted organic synthesis (MAOS) has emerged as a particularly advantageous technique due to its reduced reaction times, improved yields, and enhanced selectivity. This study reports the efficient synthesis of 3,7 dihydroxyxanthone through the reaction of 2,5-dihydroxybenzoate and resorcinol, using Eaton’s reagent and zinc chloride (ZnCl2) as catalyst under MAOS conditions. The structures of the resulting compounds were analyzed using Fourier-transform infrared spectroscopy (FTIR) and proton nuclear magnetic resonance (1H-NMR). The efficiency of the catalytic systems was evaluated based on the product yields. The Eaton reagent catalysis resulted in a 9.38% yield of a red solid, while the ZnCl2-catalyzed reaction produced a significantly higher yield of 23.07%, with the product appearing as an orange solid. These results demonstrate that Zn
References
Journal of Molecular Structure, 1177; 215–228
Aye, A., Y.-J. Song, Y.-D. Jeon, and J.-S. Jin (2020). Xanthone Suppresses Allergic Contact Dermatitis In Vitro and In Vivo. International Immunopharmacology, 78; 106061
Bosson, J. (2023). Scope and Limitations of the Preparation of Xanthones Using Eaton’s Reagent. Turkish Journal of Chemistry, 47(6); 1420–1428
Castanheiro, R. A. P., M. M. M. Pinto, S. M. M. Cravo, D. C. G. A. Pinto, A. M. S. Silva, and A. Kijjoa (2009). Improved Methodologies for Synthesis of Prenylated Xanthones by Microwave Irradiation and Combination of Heterogeneous Catalysis (K10 Clay) with Microwave Irradiation.Tetrahedron, 65(19); 3848–3857
Dharavath, R., N. Nagaraju, M. R. Reddy, D. Ashok, M. Sarasija, M. Vijjulatha, T. Vani, K. Jyothi, and G. Prashanthi (2020). Microwave-Assisted Synthesis, Biological Evaluation and Molecular Docking Studies of New Coumarin-Based 1,2,3-Triazoles. RSC Advances, 10(20); 11615–11623
Fatmasari, N., Y. S. Kurniawan, Jumina, C. Anwar, Y. Priastomo, H. D. Pranowo, A. K. Zulkarnain, and E. N. Sholikhah (2022). Synthesis and In Vitro Assay of Hydroxyxanthones as Antioxidant and Anticancer Agents. Scientific Reports, 12(1); 1535
Feng, Z., X. Lu, L. Gan, Q. Zhang, and L. Lin (2020). Xanthones, a Promising Anti-Inflammatory Scaffold: Structure, Activity, and Drug Likeness Analysis. Molecules, 25(3); 598
Gawande, M. B., S. N. Shelke, R. Zboril, and R. S. Varma (2014). Microwave-Assisted Chemistry: Synthetic Applications for Rapid Assembly of Nanomaterials and Organics. Accounts of Chemical Research, 47(4); 1338–1348
Genovese, S., S. Fiorito, M. C. Specchiulli, V. A. Taddeo, and F. Epifano (2015). Microwave-Assisted Synthesis of Xanthones Promoted by Ytterbium Triflate. Tetrahedron Letters, 56(6); 847–850
Huang, Q., Y. Wang, H. Wu, M. Yuan, C. Zheng, and H. Xu (2021). Xanthone Glucosides: Isolation, Bioactivity and Synthesis. Molecules, 26(18); 5575
Kurniawan, Y. S., N. Fatmasari, H. D. Pranowo, E. N. Sholikhah, and Jumina (2024). Investigation on Anticancer Agent Against Cervical and Colorectal Cancer Cell Lines: One-Pot Synthesis, In Vitro and In Silico Assays of Xanthone Derivatives. Journal of Applied Pharmaceutical Science, 14(3); 145–153
Liu, J., H. Bao, H. Wang, Q. Luo, J. Zuo, Z. Liu, S. Qiu, X. Sun, and X. Liu (2019). Synthesis of Xanthone Derivatives and Anti-Hepatocellular Carcinoma Potency Evaluation: Induced Apoptosis. RSC Advances, 9(70); 40781–40791
Liu, J., J. Zhang, H. Wang, Z. Liu, C. Zhang, Z. Jiang, and H. Chen (2017). Synthesis of Xanthone Derivatives and Studies on the Inhibition Against Cancer Cells Growth and Synergistic Combinations of Them. European Journal of Medicinal Chemistry, 133; 50–61
Lu, Y., T. Guan, S. Wang, C. Zhou, M. Wang, X. Wang, K. Zhang, X. Han, J. Lin, Q. Tang, C. Wang, and W. Zhou (2023).
Novel Xanthone Antibacterials: Semi-Synthesis, Biological Evaluation, and the Action Mechanisms. Bioorganic & Medicinal Chemistry, 83; 117232
Martina, K., G. Cravotto, and R. S. Varma (2021). Impact of Microwaves on Organic Synthesis and Strategies Toward Flow Processes and Scaling Up. The Journal of Organic Chemistry, 86(20); 13857–13872
Maurya, M. R., S. Dhaka, and F. Avecilla (2015). Oxidation of Secondary Alcohols by Conventional and Microwave-Assisted Methods Using Molybdenum Complexes of ONO Donor Ligands. New Journal of Chemistry, 39(3); 2130–2139
Pinto, M. M. M., A. Palmeira, C. Fernandes, D. I. S. P. Resende, E. Sousa, H. Cidade, M. E. Tiritan, M. Correia-da Silva, and S. Cravo (2021). From Natural Products to New Synthetic Small Molecules: A Journey through the World of Xanthones. Molecules, 26(2); 431
Praceka, M. S., S. Megantara, R. Maharani, and M. Muchtaridi (2021). Comparison of Various Synthesis Methods and Synthesis Parameters of Pyrazoline Derivates. Journal of Advanced Pharmaceutical Technology and Research, 12(4); 321–326
Ramakrishnan, S., N. Mad Nasir, J. Stanslas, A. I. Faisal Hamdi, M. A. Mohammad Latif, and F. F. Baharuddin (2023).
One-Pot Two-Component Synthesis of Halogenated Xanthone, 3-O Substituted Xanthone, and Prenylated Xanthone Derivatives as Aromatase Inhibitors. Results in Chemistry, 5; 100789
Resende, D. I. S. P., P. Pereira-Terra, A. S. Inacio, P. M. Da Costa, E. Pinto, E. Sousa, and M. M. M. Pinto (2018). Lichen Xanthones as Models for New Antifungal Agents. Molecules, 23(10); 2617
Rovani, S., J. J. Santos, P. Corio, and D. A. Fungaro (2018).Highly Pure Silica Nanoparticles with High Adsorption Capacity Obtained from Sugarcane Waste Ash. ACS Omega, 3(3); 2618–2627
Sulistyowaty, M. I., D. Setyawan, P. P. M. Prameswari, R. J. K. Susilo, T. Amrillah, E. Zaini, and S. A. H. Zidan (2024). A Comparison Study between Green Synthesis of Microwave Irradiation and Solvent Evaporation Methods in The Formation of p-Methoxycinnamic Acid-Succinic Acid Cocrystals. Science and Technology Indonesia, 9(3); 629–636
Tiwari, G., A. Khanna, V. K. Mishra, and R. Sagar (2023). Recent Developments on Microwave-Assisted Organic Synthesis of Nitrogen- and Oxygen-Containing Preferred Heterocyclic Scaffolds. RSC Advances, 13(47); 32858–32892
Verbanac, D., S. C. Jain, N. Jain, M. Chand, H. Čipčić Paljetak, M. Matijašić, M. Perić, V. Stepanić, and L. Saso (2012). An Efficient and Convenient Microwave-Assisted Chemical Synthesis of (Thio)Xanthones with Additional In Vitro and In Silico Characterization. Bioorganic & Medicinal Chemistry, 20(10); 3180–3185
Wu, J., J. Dai, Y. Zhang, J. Wang, L. Huang, H. Ding, T. Li, Y. Zhang, J. Mao, and S. Yu (2019). Synthesis of Novel Xanthone Analogues and Their Growth Inhibitory Activity Against Human Lung Cancer A549 Cells. Drug Design, Development and Therapy, 13; 4239–4246
Xochicale-Santana, L., C. C. Vidyasagar, B. M. Muñoz-Flores, and V. M. J. Pérez (2021). Chapter 15 - Microwave Assisted Organic Syntheses (MAOS): The Green Synthetic Method. In B. Kharisov and O. Kharissova (Eds.), Handbook of Greener Synthesis of Nanomaterials and Compounds. Elsevier, pages 491–542
Yadav, D. K., P. Kaushik, Pankaj, V. S. Rana, D. Kamil, D. Khatri, and N. A. Shakil (2019). Microwave Assisted Synthesis, Characterization and Biological Activities of Ferrocenyl Chalcones and Their QSAR Analysis. Frontiers in Chemistry, 7; 814
Yuanita, E., N. Hidayah, M. R. Umami, B. N. S. Ningsih, M. Ulfa, Sudirman, and N. K. T. Dharmayani (2024). Simple Synthesis and Characterization of Sulfonate Ester-Substituted Hydroxyxanthone. AIP Conference Proceedings, 3055(1)
Yuanita, E., B. K. Jannah, M. Ulfa, Sudirman, B. N. S. Ningsih, and N. K. T. Dharmayani (2023). C-Prenylation of 1,3-Dihydroxyxanthone: Synthesis, Characterization and Antibacterial Activity. Acta Chimica Asiana, 6(1); 279–286
Yuanita, E., H. D. Pranowo, D. Siswanta, R. T. Swasono, Mustofa, A. K. Zulkarnain, J. Syahri, and Jumina (2018). One-Pot Synthesis, Antioxidant Activity and Toxicity Evaluation of Some Hydroxyxanthones. Chemistry and Chemical Technology, 12(3); 290–295
Authors

This work is licensed under a Creative Commons Attribution 4.0 International License.